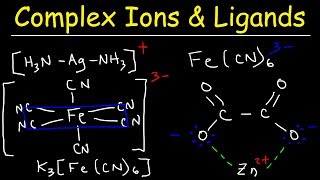
Web Reference: Apr 12, 2023 · A complex ion forms from a metal ion and a ligand because of a Lewis acid–base interaction. The positively charged metal ion acts as a Lewis acid, and the ligand, with one or more lone pairs of electrons, acts as a Lewis base. Many slightly soluble ionic solids dissolve when the concentration of the metal ion in solution is decreased through the formation of complex (polyatomic) ions in a Lewis acid-base reaction. Learn what a complex ion is, how it forms by co-ordinate bonds with ligands, and how to name and classify it. See examples of complex ions with different metal ions and ligands, and how their coordination numbers vary.
YouTube Excerpt: Most transition metal cations can do something interesting in solution, they can interact with specific ligands to form
Information Profile Overview
Complex Ion Formation - Latest Information & Updates 2026 Information & Biography

Details: $87M - $108M
Salary & Income Sources
Career Highlights & Achievements

Assets, Properties & Investments
This section covers known assets, real estate holdings, luxury vehicles, and investment portfolios. Data is compiled from public records, financial disclosures, and verified media reports.
Last Updated: April 4, 2026
Information Outlook & Future Earnings

Disclaimer: Disclaimer: Information provided here is based on publicly available data, media reports, and online sources. Actual details may vary.








